Reviewing topical JAK Inhibitors for hair loss
Almost every time I see a new hair loss patient, they come in asking about JAK inhibitors, usually toting articles or printouts from the Internet or social media. They have certainly received a lot of press and unquestionably there needs to be some clarification on their use.
JAK inhibitors are a groups of medications that inhibit a pathway inside cells known as the janus kinase signal transducer and activator of transcription (STAT) pathway. These kinases (enzymes) play an essential crucial role in immunity as well as in the formation of blood cells and components. When this pathway is restricted, the activation of immune cells and T-cell inflammation is overpowered, which makes it an inviting medication to treat inflammatory types of diseases.
There are currently two FDA-approved JAK inhibitors accessible for human use, but they have a steep price tag. Xelijanz (tofacitinib) is a first generation JAK inhibitor approved for treatment of rheumatoid and psoriatic arthritis. The retail cost of Xelijanz is approximately $4,075 a month. Jakafi (ruxolitinib) is another first generation JAK inhibitor and it is approved for myleofibrosis (aka osteomyelofibrosis), a rare bone marrow cancer and polycythemia vera (when the bone marrow makes too many red blood cells). The retail cost of Jakafi is more than $12,162 a month. There is a third JAK inhibitor available, oclacitinib, also a first generation JAK inhibitor, but is only approved for allergic dermatitis in canines. There are other JAK inhibitors coming down the pipe. Baricitinib is a first generation JAK inhibitor being studied for rheumatoid arthritis, psoriasis and atopic dermatitis, but is not yet approved. Additionally, there are some second generation JAK inhibitors being investigated.
There are lots of concerns over the systemic administration of JAK inhibitors because of their side effects. These side effects include serious infections, malignancies and decreased blood cell counts. Others like herpes zoster, bronchitis and urinary tract infections occur more frequently. As a result of the severe nature of some of these side effects, it is easy to see the attractiveness of topical formulations containing JAK inhibitors.
Over the years there have been much progress in knowledge of the pathology of psoriasis, vitiligo and atopic dermatitis. All are considered autoimmune skin diseases, which means topical JAK inhibitors may be a hopeful new treatment. In studies, topical JAK inhibitors seem to be safe and well-tolerated with low incidences of adverse effects. Moreover, they offer a direct anti-inflammatory effect on the skin.
Psoriasis
In five randomized controlled trials and one cohort study, topical tofacitinib and ruxolitinib were studied for the treatment of psoriasis. Unfortunately, the results only yielded limited improvement in the signs and symptoms of the disease. Additionally, in 2015 a phase 3 study of oral tofacitinib was beneficial in the treatment of psoriasis, but approval was turned down by the FDA because of safety concerns. However in 2017, oral tofacitinib was approved for the treatment of psoriatic arthritis.
Atopic Dermatitis
When used in atopic dermatitis, JAK inhibitors prevent inflammation and skin barrier dysfunction. There has been favorable outcomes in mouse model studies of atopic dermatitis with topical JAK inhibitors, lessening itching and inflammation as opposed to oral formulations that just decreased itching. Improvement of itching in atopic dermatitis is a huge therapeutic benefit (atopic dermatitis patients get very itchy!). Other studies with topical JAK inhibitors are suggestingly be advantageous to patients with atopic dermatitis because they diminish disease severity and itching by disturbing the connection to the sensory neurons that cause itch. At this time there is also an multi-centered clinical trial in progress on the use of ruxolitinib in pediatric atopic dermatitis. The drawback to topical JAK inhibitors in the treatment of atopic dermatitis is that rebound itching could occur after an abrupt discontinuation of the drug.
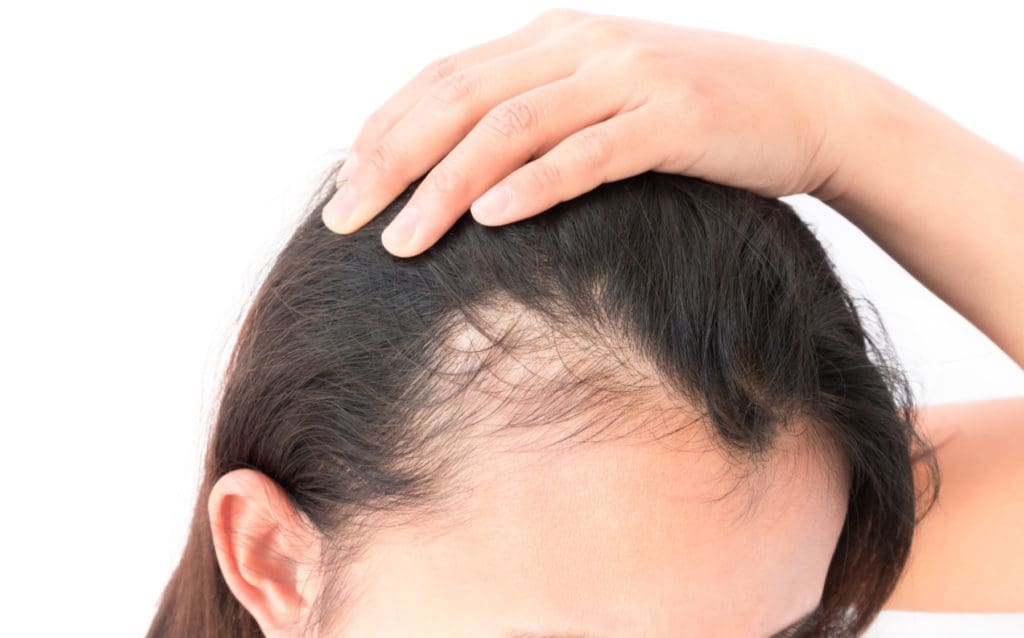
Alopecia Areata
Treatment of alopecia areata with oral JAK inhibitors have demonstrated safety and efficacy in many case reports and clinical studies, however, the evidence for an overall benefit is conflicted and limited. At this time, there are two phase 2 trials going on to assess topical tofacitinib and topical ruxolitinib in the treatment of alopecia areata. Furthermore, there were promising results in a study on pediatric alopecia areata patients, but the study was not controlled and the sample size was small.
Vitiligo
Only one study has been positive in the treatment of facial vitiligo with a topical JAK inhibitor. At this time, there is a multi-centered clinical trial on the use of topical ruxolitinib in the treatment of vitiligo.
So…do we believe the hype?